Abstract
Poster “Predicting severity of drug-induced liver injury with uncertainty”.
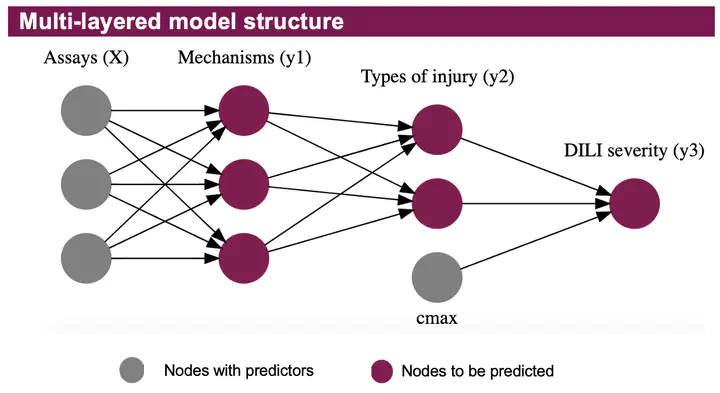
Abstract: Drug-induced liver injury (DILI) is a major cause of attrition in drug development and a common reason for withdrawing a drug from the market. Predicting clinical liver toxicity is difficult, but preclinical in vitro assays and physical/chemical properties of drugs can be used as predictors. We developed a multi-layered Bayesian model where we use assay results to predict the mechanism(s) of toxicity, use the mechanisms to predict the type of liver injury, and then combine the type of injury with the clinical dose of a drug to predict the severity of injury. The model therefore has a layered structure, enabling uncertainty to propagate through the layers. Based only on assay and physchem data, along with the clinical dose, the model enables safety pharmacologists to predict the severity, type, and mechanism of liver toxicity with good accuracy.
Poster and code are available here.